Our Research
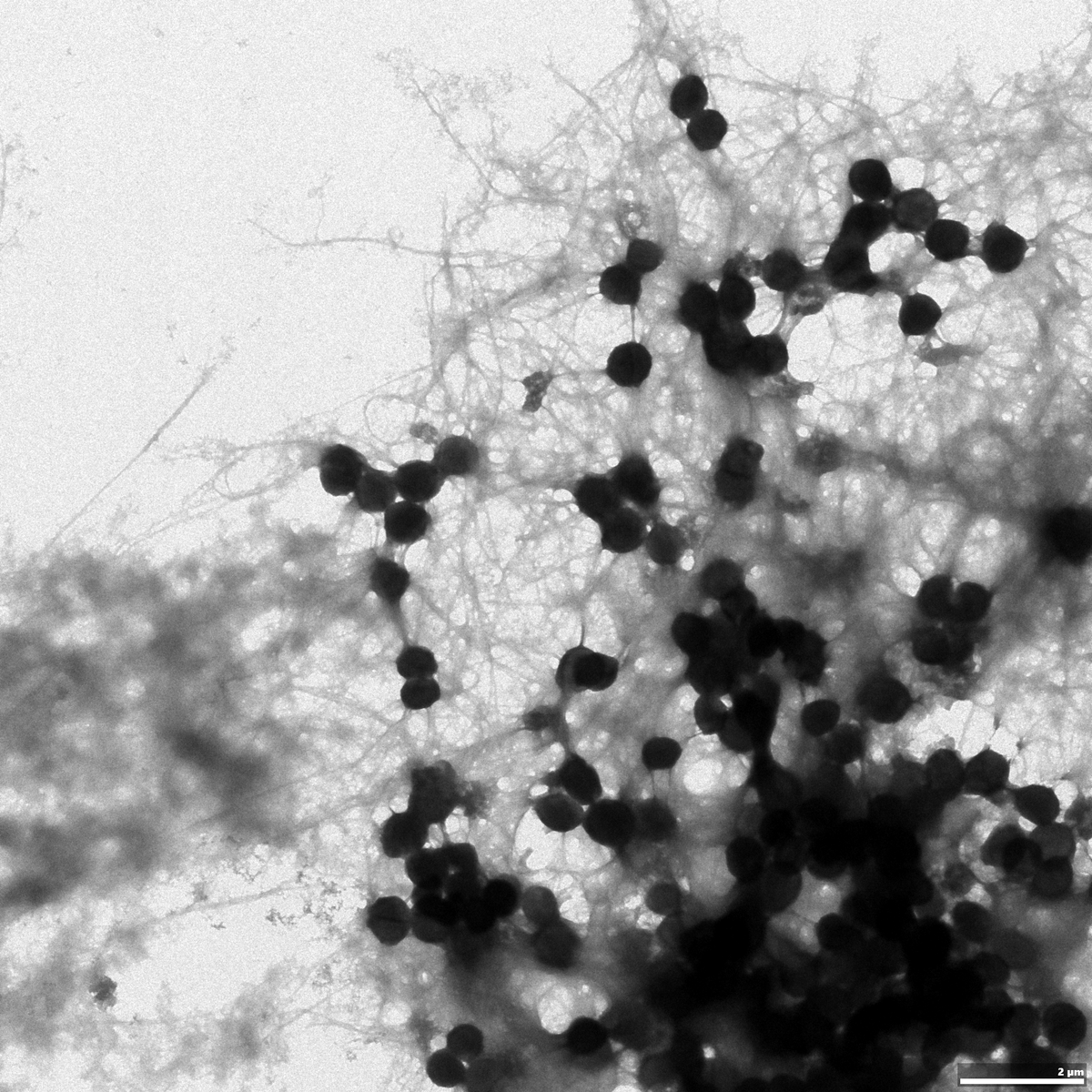
The focus of our research is functional bacterial amyloids (FuBA) found in bacterial biofilm. Functional amyloids are a unique class of amyloids with specific biological functions in living organisms, in contrast to the pathological amyloids associated with diseases such as neurodegenerative diseases. 80% of all chronic infections are either related to or caused by both biofilm formation and biofilm-mediated resistance to antimicrobial and antibiotic therapies. Biofilms are composed of macromolecules such as DNA, polysaccharides and proteins secreted from the bacteria. Self-assembly of the functional amyloid proteins in the biofilm enhances the stability of the biofilm and renders the biofilm impossible to disassemble. We use biophysical techniques such as fluorescence plate-reader assays, kinetic analysis, spectroscopic assays and electron microscopy to study the self-assembly of FuBA proteins and how this process can be modulated. Specifically we investigate functional amyloids formed by FapC (from pseudomonas) and PSMs (from Staphylococcus aureus). Our studies will establish a better understanding of the functional amyloid formation in bacterial biofilms on a molecular level. We hope to ultimately pave the way for the development of future treatments strategies against bacterial infections and their associated antimicrobial resistance.
Modulation of functional amyloid formation
Using biophysical techniques, we investigate how functional amyloid formation can be modulated by other factors. This includes changes in the physio-chemical environment such as pH, temperature, buffer components and salt, presence of other components from the biofilm (DNA or other macromolecules) and effects of different surfaces. We study the changes in aggregation kinetics and correlate this molecular events taking place during the self-assembly process, changes in aggregate morphology and changes in functionality and stability.
Structural analysis of functional amyloids
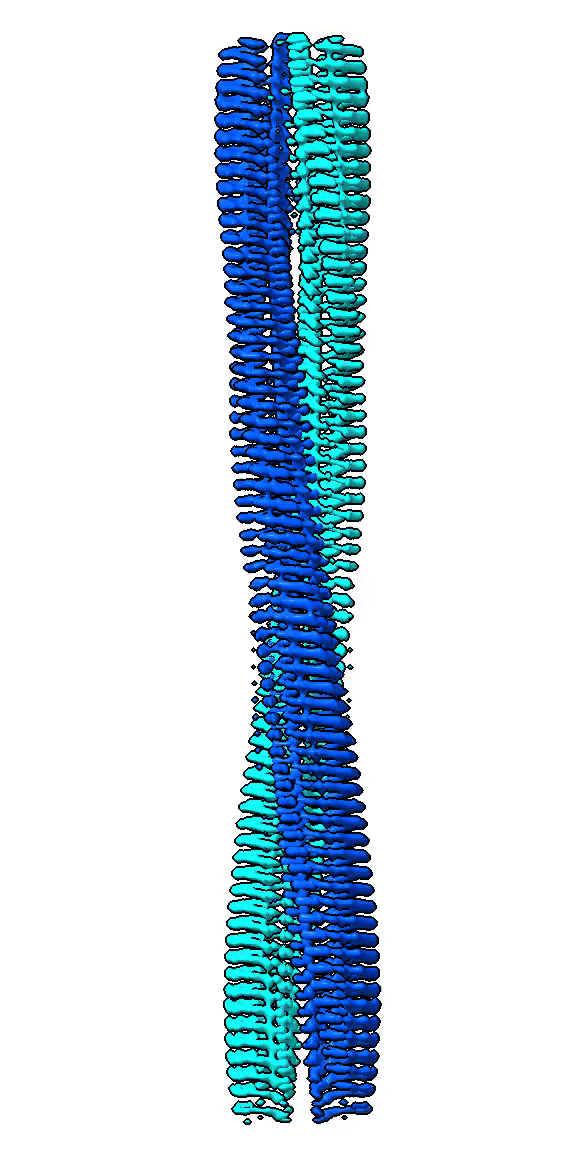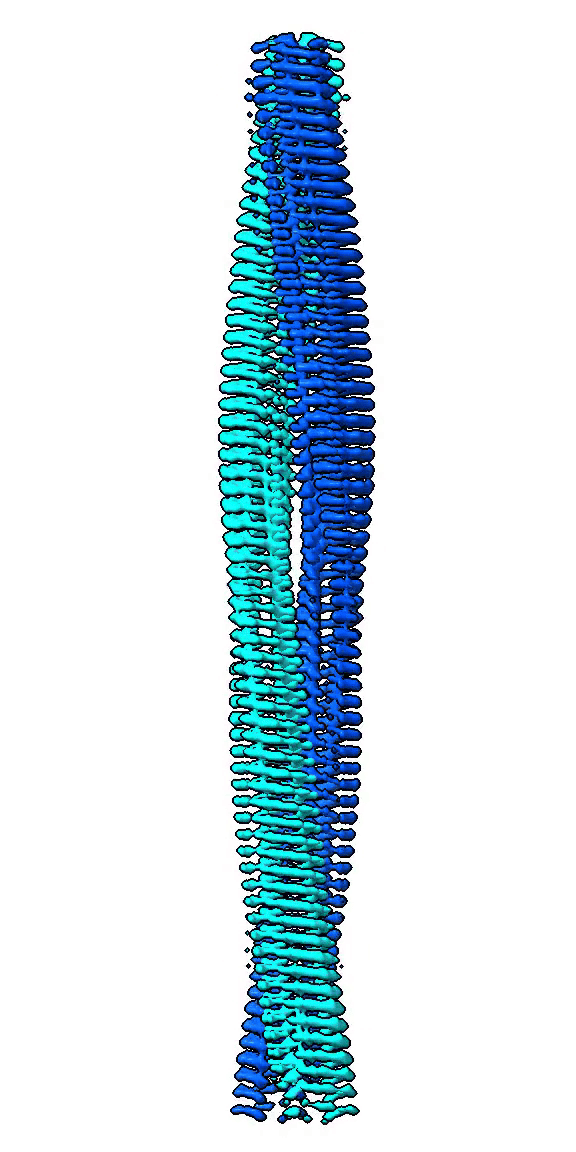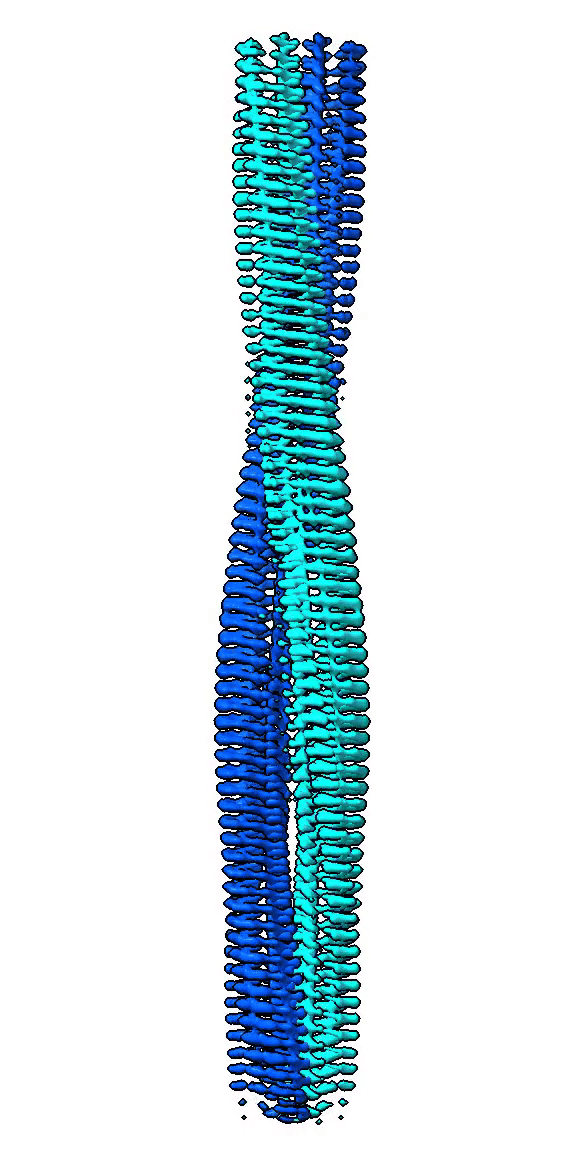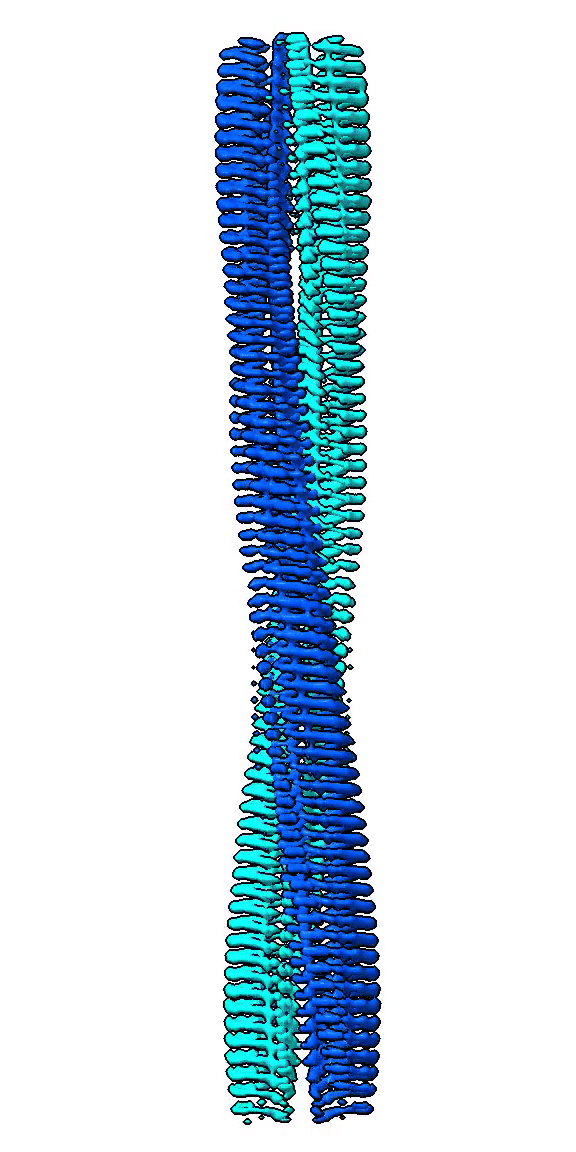
We use cryo-EM to study high-resolution structures of functional amyloid forming proteins. Based on these structures we investigate how individual amino acids contribute to the structure and stability of the structure. We also study how different structures correlate with functionality such as interactions with cell membranes through cell toxicity assays and exposure of hydrophobic patches.